^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^^

Science speechI have often boasted I could write a speech about any topic given five minutes of research. People scoff. They assume much more than the words I have just spoken. How long might that speech be? It would be whatever I learn in five minutes of research plus my opinion about the subject. Here’s where I prove myself. I have opinions to call upon and I try to make them as outrageous as possible so that people will pay attention. Otherwise, my speech will be boring and will seem to take up much more time than a boring speech should be allotted. You know the type. Blah, blah, blah, Oh look, I got a text from my cousin.
First of all, it doesn’t matter that I know nothing about the topic and, if you all agree with that assumption, then I can proceed with new and exciting information that will have zero impact upon your happy little world…unless you are well-versed in the chosen topic and become confused or incensed by my complete lack of knowledge. If you have no intimate relationship to this subject, I believe you will be Googling up a storm after hearing my speech. Either way, don’t be mad at me. It’s just a speech and won’t affect you like a massive dose of Benadryl–so, no inadvertent snoozing.
Science doodlesThe title of this week’s blog post was contributed by my husband. I was thrilled by the challenge. Let’s see. Breaking it down. Thermo has to do with temperature, maybe. Dynamic is always an exhilarating word. Chemistry is that subject I dropped in college because it was not at my usual nap time. I could easily come up with a speech in the moment, but decided to put in the stated amount of work to give myself at least as thorough an amount of knowledge as I deemed necessary to get a good start.
After short, but exhausting research, my explanation of thermodynamic chemistry is this: Shit Happens. It happens in a system where there is energy involved. Uncertainty or disorder is at hand. Chemical changes are all but certain. Stuff moves around if there is a change in the system’s environment, especially if it involves heat. With the summer we are having, this seems somewhat relevant to our lives. Allow me to give you an example of this amazing science.

Questionable decisionIt’s summer in the northern hemisphere. You’ve gone outside in the afternoon to mow your lawn. You are (whether you are aware of it or not) a magnificent, highly complex system. The sun is shining–not a cloud in the sky. It’s 97 degrees. It’s been a month since you last mowed, so the grass is gnarly high on your little patch of turf. Your body-system has been well-fed on yesterday’s leftover chili and has plenty of energy to expend. Are you with me so far?
You crank up the gas-powered lawn mower (a system in itself) and start hacking away at the grass. First in short bursts since it’s pretty difficult to get it going through the thatchy growth. Then, after working up a head of steam, you get into your groove. Maybe you have BTS or Taylor Swift playing on your I Phone to help give you some momentum. Your system is creating energy (from the chili) and using energy (for the mowing) at the same time. You get hot. You sweat. You sweat some more. You begin to deplete the water in your system. Your internal body temperature increases to a degree which medical professionals deem “start praying” level. The sodium (and electrolytes-whatever they are) level in your blood (this is the chemistry part) depletes drastically. You shout “Get me some Gatorade!” but no body hears you.

Thermodynamic stressNow, all you sweaty beasts out there should be aware of the need for sodium in the human body. It’s what facilitates nerve function and muscle contraction. Without it, you are just a floppy meat sack. The amount of sodium required for maintaining a human operational system is smallish, and that is why your doctor tells you to cut back on the potato chips. Too much salt changes your blood chemistry and does a number on your blood pressure. Total annihilation of your sodium supply through massive sweating is detrimental to your continued moving about (i.e. the activity coefficient of your ideal solution). In any event, your body does not appreciate losing all it’s water either. It’s dicey, this body chemistry stuff.
Your system, once subjected to your poor decision to mow the lawn in the 97 degree heat, will start to send you internal messages like headache, nausea or weariness (heat exhaustion). If your brain is still in working order, you quit for the day and eat a prudent number of potato chips with your Gatorade–system restored. If yours is a stubborn human system, and you press on, your brain function begins to deteriorate and you become really stupid and combative, and collapse in the gnarly high grass (heat stroke). Your neighbor, who has been tsk-tsking your questionable chore schedule, calls 911. Thermodynamic chemistry is important. Remember that the next time you are determined to go outside during a heat wave to knock something off your to-to list.
So, to restate my example into an attractive and comprehensive chart:
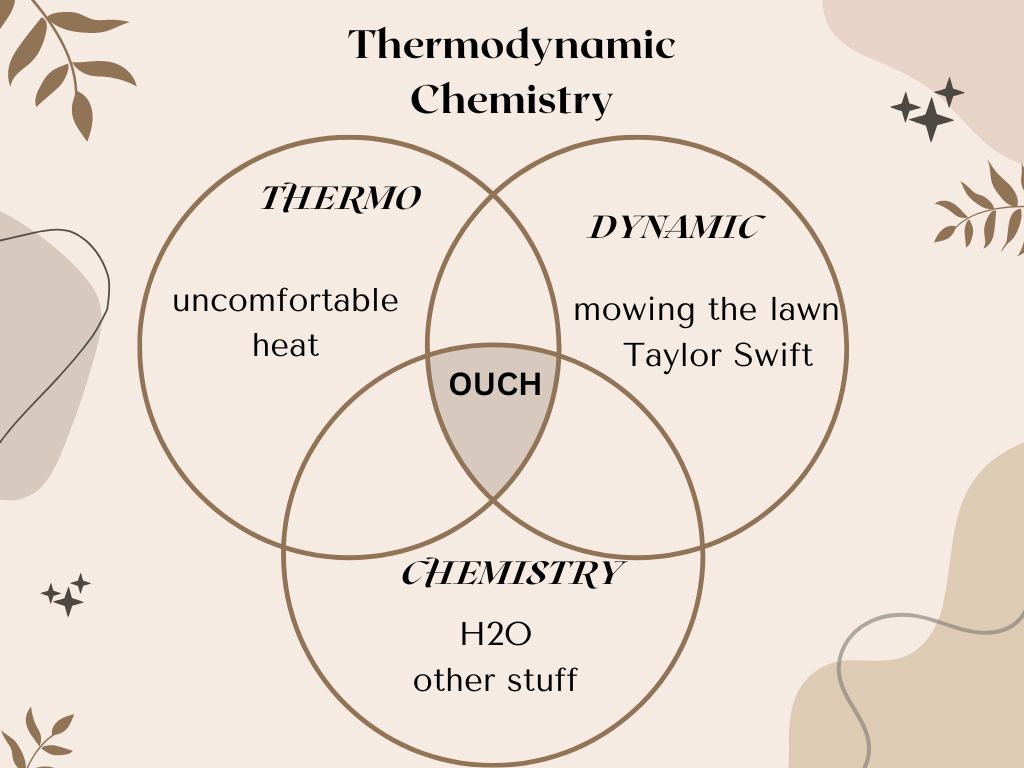
My first thought on hearing the term thermodynamics was a long-ago lesson about steam engines and the transfer of energy into work. I think we all can reach the consensus that this stuff is quite dull until you add the chemistry component. Yes, yes, advances in engineering and civilization and all that, but understanding how this science-y stuff works in your own body brings the concept right to your door and saves you from the disaster of dehydration and accompanying deleterious disorders.

Fav systemThere are, of course, other types of systems in the universe which are of importance to every one of us. The universe for one. The solar system has the word system right there in the name and my home, Earth, is one of my favorites. In my exhausting research, I found that every system could have chemical reactions given changes to the environment. Does any of this sound familiar? Just like your own human system, mowing the grass and failing miserably, the planet is being warmed and may soon keel over in much the same fashion. But this isn’t meant to be a rant on climate change, just a small reminder that thermodynamic chemistry affects us all, even if we refuse to acknowledge it.
Perhaps there should be an awareness campaign. Some might say thermodynamic chemistry should be taught starting in the first grade (Kindergarten should still be fun) and continued until we all become aware of the forces that drive our many systems. I’m up for an annual holiday and accompanying celebrations with public demonstrations of Raoult’s Law and other important concepts. Think how different life would be if we all paid attention to the intricacies of scientific knowledge. We could call it Ideal Solution Day. And that would make me very happy as the name has a double meaning, which, if you had Googled thermodynamic chemistry, you would be nodding in agreement.

Chillin'Now take a deep breath and upon exhaling, recite Boyle’s Law, because without it’s described action, you could not live. It’s obvious that most of us go through our days without the slightest notion of the majority of these chemical reactions and are none the worse for wear. We try to lounge around with a cool drink on 97 degree days because we instinctively perceive that our bodies don’t operate functionally in the freakishly dangerous heat. May your own body system maintain equilibrium and happiness as you drive the dynamic meat sack through your days. Remember though, that there’s science stuff going on in there and respect the fact that you are not immune to change. As they say in thermodynamic chemistry circles…Baby, it’s hot out there!
Keep your cool and your sodium,
Cheryl
Learn more stuff in Quote of the Week
Disclaimer: The writer/editor of this blog has no idea what she’s talking about, but should be forgiven for the sake of light entertainment.




What if I don’t like the taste of Gatorade
Gosh, who does? It tastes like worms crawled through it and water from my gutters drained into it. It’s known for its disgusting taste and that’s probably why it’s good for you. Stick to water and a few Doritos and you should be fine all summer long.
What a delightful read – I try to live by the adage, you don’t have to understand to respect….in this situation, science – for more interesting science education try Flanders and Swann’s musical Boyle’s Law – https://www.youtube.com/watch?v=VnbiVw_1FNs (Flanders and Swann are a beloved British institution of silly songs for those of us growing up in the ’50s and ’60s)
I clicked on your link and thank you for referring me to these impressive fellows. Now I want to write a musical about science-y things. A whole new world awaits. I am newly enlightened!
And another piece written I. Such a way that helps me know that it’s ok not to give a f*ck! ????
Well done
So happy to have contributed to your overall not-give-a-f**kedness!
Cheryl, you met your husband’s challenge to the max. Kudos to you for another fun and informative blog post. Stay cool. It becomes you.
Mr. Ethel sure thought I’d be stumped on this one, but as I say, even the most boring stuff can be jazzed up a bit for general consumption. Thanks for reading!
Nice paper, but is Wikipedia an acceptable pier-reviewed citation? Also, what is the reference to Boyle’s law for? Boyle’s law is a gas law, stating that the pressure and volume of a gas have an inverse relationship. What does that have to do with Raoult’s law, ideal solutions, and thermodynamic chemistry?
Pepe Raoult, Francois-Marie’s brother
Dear Pepe, You must be pretty freaking old by now. Is your brain swimming in a jar somewhere? I only said you couldn’t live without the action of Boyle’s Law, which is true. Everything has to do with thermodynamic chemistry. And yes, my peers have reviewed Wikipedia many times, so it’s a legit resource when writing poorly informed science papers, which this wasn’t…a science paper that is.
And now I can’t stop thinking about Krebs Cycle (which has nothing to do with bicycles).
So many good science jokes!
Oh my, and to think I knew you when! My sojourn into chemistry lasted for just one class when a Junior in high school, after our teacher said we had to memorize the periodic table. I took Drama in its place.
Yes, good choice. Yet drama and chemistry often go hand in hand. At least that’s what I’m told.